This is the ninth article in a series where I examine the structural conditions that would need to change for Europe to function as a genuinely independent strategic actor.
In the previous article I established that three supply chain vulnerabilities, ammunition, pharmaceuticals, and food security, are one structural condition, with upstream chemistry and critical input production concentrated in adversarial or conditionally reliable territory, shared across all three chains simultaneously.
In this article I trace the pharmaceutical supply chain that European forces would actually require under conflict conditions, not what European healthcare systems currently stock, but what battlefield wound infections with the resistance profiles documented in Ukraine would demand, from the finished injectable dose back to the elemental inputs that production depends on.
Answering that technical question required me to establish what planning standard I must apply, and why.
The answer to that question forced reclassifications that European strategic thinking has not generally applied to these relationships. This includes reclassifying Turkey, which controls 72 to 75 percent of global boron reserves through a state enterprise and has imposed unilateral export controls on boron in three consecutive years while remaining a nominal NATO ally. And it includes the United States, where a single 2016 transaction placed commercial control of the most critical antibiotic classes at Steps 2, 3, and 4 of the treatment ladder in the hands of one company whose supply reliability depends on US executive disposition toward Europe at the moment of need.
What I found, working through the supply chain systematically, is that the European pharmaceutical manufacturing that exists is in significant parts a final assembly operation for inputs that come from somewhere else entirely, and that the planning standard the democratic obligation toward human life requires produces a demand profile that the supply chain, as it currently stands, cannot meet.
What conflict demands of a democratic state
I served in the Swiss armed forces and later volunteered for international armed peacekeeping missions in conflict-torn regions. There is a fundamental principle that is part of Swiss military training and military training for peacekeeping missions, that is simultaneously a rule of engagement, and a statement of values. In combat, only the least necessary degree of force may be used against an adversary, which in practice means that you must not fire in bursts or fire consecutive shots at an individual, since every bullet fired must be justifiable.
As soon as an adversary stops fighting, whether due to incapacitation resulting from being wounded or a decision to surrender, they are no longer considered an adversary, and the obligation toward them changes at that moment. From that moment on, one is obligated to treat the surrendered with dignity, or to do everything in one’s power to save a wounded individual’s life. This reflects the formalization in military doctrine of the principle that the value of a human life does not depend on which side of a conflict that individual is on.
That recognition is what human rights mean when they are treated as non-negotiable. A democratic society and government that accepts human rights as foundational, acknowledging that each and everyone of us have an intrinsic value, then it accepts a constraint on its conduct that does not suspend when the conduct becomes difficult or costly. The principle of least necessary force and the obligation toward the wounded are two expressions of the same foundation. You apply no more force than necessary, and you preserve life where preservation is possible, because the alternative is to treat human life as having conditional value, which is the one thing a democracy premised on human rights cannot accept without undermining the premise.
The obligation toward your own soldiers follows from the same foundation. When a democratic government sends its citizens into conflict, it inherits a relationship with those people that is not a commercial contract. They accepted risk on behalf of the society that sent them. The minimum the society owes in return is that it does not create avoidable conditions that turn a survivable wound into a death sentence. The obligation is not to guarantee survival, because no honest government can promise that. It is not to allow a preventable death to occur because a decision made in peacetime failed to account for what conflict would actually require.
A soldier who develops a wound infection and dies because the right antibiotic was unavailable died in a hospital bed from a treatable condition because the supply chain that should have been established was lacking. Although he was exposed to the bacteria on the battlefield, it was the failure of the government and society that cost him his life. One must understand the difference between both causes of death before a supply chain analysis of this kind can be viewed with its full weight.
The planning standard, and how I arrived at it
Before I could assess any supply chain, I had to decide what I am assessing it against. That question sounds technical, but it is the question on which everything else depends, because planning for the most likely outcome and planning for the worst case produce completely different requirements, investment figures, and institutional mandates.
While researching the pharmaceutical supply chain, I reflected on my time and experiences in uniform, as well as what I consider to be inherently true about the role of man and society, requiring me to answer questions such as what the purpose of society is, what role does the individual play within it, what should the relationship between an individual and society look like, and what obligations do both sides have toward one another?
Explaining these views and beliefs in detail would go beyond the scope of this article. For the purposes of this article, it should suffice to acknowledge that every individual has their purpose, values, convictions, beliefs, ideals, expectations, and priorities, and that no individual has the right to impose their will on another. Even when individuals form groups to advance their interests, the same principle applies, that no one may impose their will on another. This is the foundation of an open and free democratic society that pursues and forms consensus on societal, social, religious, and political concerns.
Once we acknowledge this as stated, then society’s responsibility toward the individual who is sent into battle becomes crystal clear, and with it what scenario we must assess the pharmaceutical supply chains against.
Planning for the worst case, you arrive at requirements that are larger, more expensive, and more difficult to form a consensus politically. The justification depends entirely on whether the consequences of getting it wrong make worst-case planning logically necessary rather than merely cautious. The argument for necessity is straightforward. While the error of over-preparation, building capacity that exceeds demand, stockpiling drugs that are not used, costs money, but that error is recoverable, the error of under-preparation, the last-resort drug is unavailable when the patient who needs it presents, is irrecoverable.
You cannot treat that patient retrospectively. When one error is recoverable and the other is not, and when there is a democratic obligation to the person who bears the cost of the irrecoverable error, the planning standard is determined. It is the logical consequence of accepting both the asymmetry and the obligation simultaneously.
While I arrived at that conclusion through the ethical and values-based logic described above, there is also another side to it. A few years ago, I read an article by the Jerusalem Institute for Strategy and Security (JISS) written by Prof. Col. (ret.) Gabi Siboni and Brig. Gen. (ret.) Yuval Bazak titled “The IDF “Victory Doctrine”: The Need for an Updated Doctrine”. It states the IDF's (Israeli Defense Forces) strategy verbatim: "Preparing the military for war requires that reasonable worst-case scenarios be taken into account. Israel does not and will not have the ability to anticipate the circumstances and conditions of the next war. Therefore, it always must prepare for the reasonable worst-case scenario. Otherwise, at the moment of truth, it could find itself vulnerable.", which was formalized under conditions more demanding than Europe has faced in living memory, as Israel has been in war since it was founded.
Whether you derive that principle from the asymmetry of irreversible consequences, from the democratic governments obligation toward citizens in service, or from the operational experience of a military system tested repeatedly, you arrive at the same planning standard. The convergence of those three routes on the same answer is the strongest possible justification for applying it here.
Applying it to European pharmaceutical supply chains means asking at every node whether the supply can be relied upon under the conditions where it matters most.
Reframing the dependencies - what the planning standard reveals
Applying that question consistently, without adjusting the standard to make certain answers more comfortable, produced three reframings that I want to state before the supply chain analysis, because the supply chain data only carries its full weight if you have accepted the framework that makes it significant.
The first concerns Turkey. Turkey controls 72 to 75 percent of global boron reserves through Eti Maden, a state-owned enterprise established in 1935. Boron trioxide, the mineral compound derived from those reserves, is the defining ingredient in the specific type of glass required for injectable pharmaceutical vials. Every single injectable antibiotic used in the treatment ladder requires a vial made from this glass. There is no regulatory substitute under either US or European drug safety frameworks. Turkey is a NATO member, which affects the probability of supply disruption compared to China or Russia, but NATO membership does not answer whether Turkish government decisions about export controls on boron will align with European pharmaceutical security needs under the conditions that make those decisions matter most.
The empirical answer is available and does not require speculation. Turkey imposed unilateral export taxes on boron in 2021, causing prices to spike 22 percent within six months. In 2022, Turkey imposed further export quotas, producing another 22 percent price increase and causing Schott AG, Germany's largest pharmaceutical glass manufacturer and the world's leading vial producer, to reduce output at its German plants by 12 to 15 percent. In 2023, Turkey tightened controls again to prioritize domestic industrial needs.
These decisions were made by a state enterprise acting in Turkey's economic interest, which is entirely appropriate from Turkey's perspective. They were made without reference to European pharmaceutical security requirements, because no Turkish institutional obligation extends to those requirements. Alliance membership is a military and political commitment, and it does not change the geology, the state ownership structure, or the decision calculus of a government managing a strategic asset in its own interest.
The structural implication is not specific to Turkey. It is one of this article's most important contributions to the series argument. Formal alliance membership is not a substitute for physical production capability. A planning framework that applies worst-case scrutiny to adversarial suppliers but exempts nominal allies from the same analysis is applying a political preference dressed as analysis. If the planning standard requires that supply be reliable under conflict conditions, then every supply node must be assessed against that standard regardless of the political relationship. Turkey's boron and China's pharmaceutical precursors sit in the same risk category for European strategic independence purposes, for different reasons, at different risk levels, but both are dependencies whose terms are not within European control.
The second reframing concerns the United States of America. In 2016, Pfizer acquired from AstraZeneca the commercial rights outside the United States to three drugs in a single transaction, including ceftazidime-avibactam (a combination antibiotic for carbapenem-resistant infections), meropenem's commercial distribution, and aztreonam-avibactam's development rights. These are the drugs that cover 80 to 100 percent of the triage requirement for the most resistant infections at Steps 2, 3, and 4 of the treatment ladder. Under normal conditions, this is an unremarkable feature of the global pharmaceutical market. Under conflict conditions coinciding with US-European political friction, it creates a structural exposure, where European access to the most critical antibiotic classes depends on Pfizer's continued willingness to supply on terms the US executive cannot redirect.
The basis for this reclassification is the pattern the Biden administration has made visible. During the COVID-19 pandemic, the Defense Production Act, a US law that allows the government to direct private companies to prioritise domestic needs, was invoked to redirect pharmaceutical supply regardless of existing international agreements.
And the current administration under POTUS Trump has applied direct executive pressure to private companies in domains that carry no strategic weight at all. When a television host's commentary drew presidential displeasure, the administration pressured the network to take him off the air. He was reinstated within a week when viewer cancellations and public pressure made the commercial cost too large to sustain. The reversal does not undo what the incident reveals. An administration willing to direct private commercial decisions over a late-night television programme has demonstrated that this is a tool it will reach for. The planning question is what that same tool produces when applied to something of real strategic value at a moment when US and European interests are not aligned.
No US administration has yet directed a pharmaceutical company to withhold medicine from an allied nation. That distinction should be stated plainly, but the planning standard this article applies does not ask whether a harm has already occurred. It asks whether a realistic path to that harm exists that Europe cannot close on its own. A single US company holding commercial control over the drugs covering the most resistant infections in the treatment ladder, under both democrat and republican administrations that has both the legal tools and the demonstrated willingness to direct private companies, and as in the case of the current administration, which has treated allied relationships as subject to political conditions, creates an exposure that worst-case planning must address.
The broader pattern of conditional security guarantees, trade leverage applied against European partners, and the publicly stated position on Greenland shows that the institutional limits on US executive action that previously made some of these behaviors practically unthinkable have shifted. Future administrations, regardless of party, inherit precedents that have been set by what the institutional framework has been shown to permit and what any administration now demonstrably can do.
The United States remains a reliable, aligned partner across most pharmaceutical supply chains. The reclassification is specific to assets where a US-headquartered company holds single-source commercial control and where the US executive has demonstrated the capacity and willingness to direct private companies against allied interests. For those specific assets, worst-case planning requires treating US commercial control as a conditional dependency rather than an unconditional one.
The third reframing concerns the planning scenario itself. The worst case is the outcome against which the planning standard, derived from the democratic obligation and the asymmetry of irreversible error, requires Europe to be prepared. That worst case is a multi-front conflict extending beyond twelve months, without US forces or US pharmaceutical supply, against an adversary whose military approach is calibrated to convert as many wounded soldiers as possible into logistical burdens rather than returning them to active service. The patient volumes that scenario generates, and the infection profiles those patients carry, produce drug requirements that bear almost no relationship to what European pharmaceutical supply chains currently deliver.
The treatment ladder, and what the worst-case scenario requires
The Ukraine war provides the only direct reference data available for projecting European conflict casualty and infection patterns. That data is the outcome of inadequate medicine meeting a pathogen that adapted to inadequate treatment, in a healthcare system already characterized by high antibiotic use and elevated resistance. Three compounding distortions in the Ukrainian data matter for any European projection.
Ukrainian physicians acknowledged prescribing lower antibiotic doses than guidelines recommend, in order to conserve dwindling supplies. Giving less than the required dose creates exactly the selection pressure that accelerates resistance. Susceptible bacteria are killed, but resistant mutants survive and proliferate. The result is that the 84.6 percent multidrug-resistant rate in wound isolates documented in peer-reviewed research from September 2025 is not purely the product of the battlefield environment. Partly, it is the product of the supply system itself operating under resource pressure.
The 30 percent of cases that proved untreatable under Ukrainian conditions reflects the absence of last-resort antibiotics, not their ineffectiveness, and the documented pattern of antibiotic misuse in Ukraine before the conflict, with every second patient taking antibiotics who did not need them, every third using them for viral infections where they have no effect, had already elevated resistance burdens beyond what European healthcare systems would typically produce.
The implication for European planning is that the Ukrainian figures are the outcome of a specific combination of pre-existing burden, supply constraint, and system failure under pressure. A European conflict would begin from a lower resistance baseline in most countries for some forces, but two things close that gap quickly. Resistant bacteria spread between patients in shared field hospitals and casualty collection points regardless of which country's forces they came from. Once a resistant strain is present in a shared treatment facility, soldiers from low-resistance populations are exposed just as readily as those from high-resistance ones. The second factor is that the supply chain Europe currently has is so far below any of the coverage floors described further below that the same supply pressure which drove Ukrainian physicians to underdose their patients would apply to European forces in a conflict. Soldiers would receive insufficient treatment for the same reason Ukrainian soldiers did, because the right medicines were not there. Where a European conflict draws on forces from Romania, Bulgaria, or Greece, the starting point would not be lower at all.
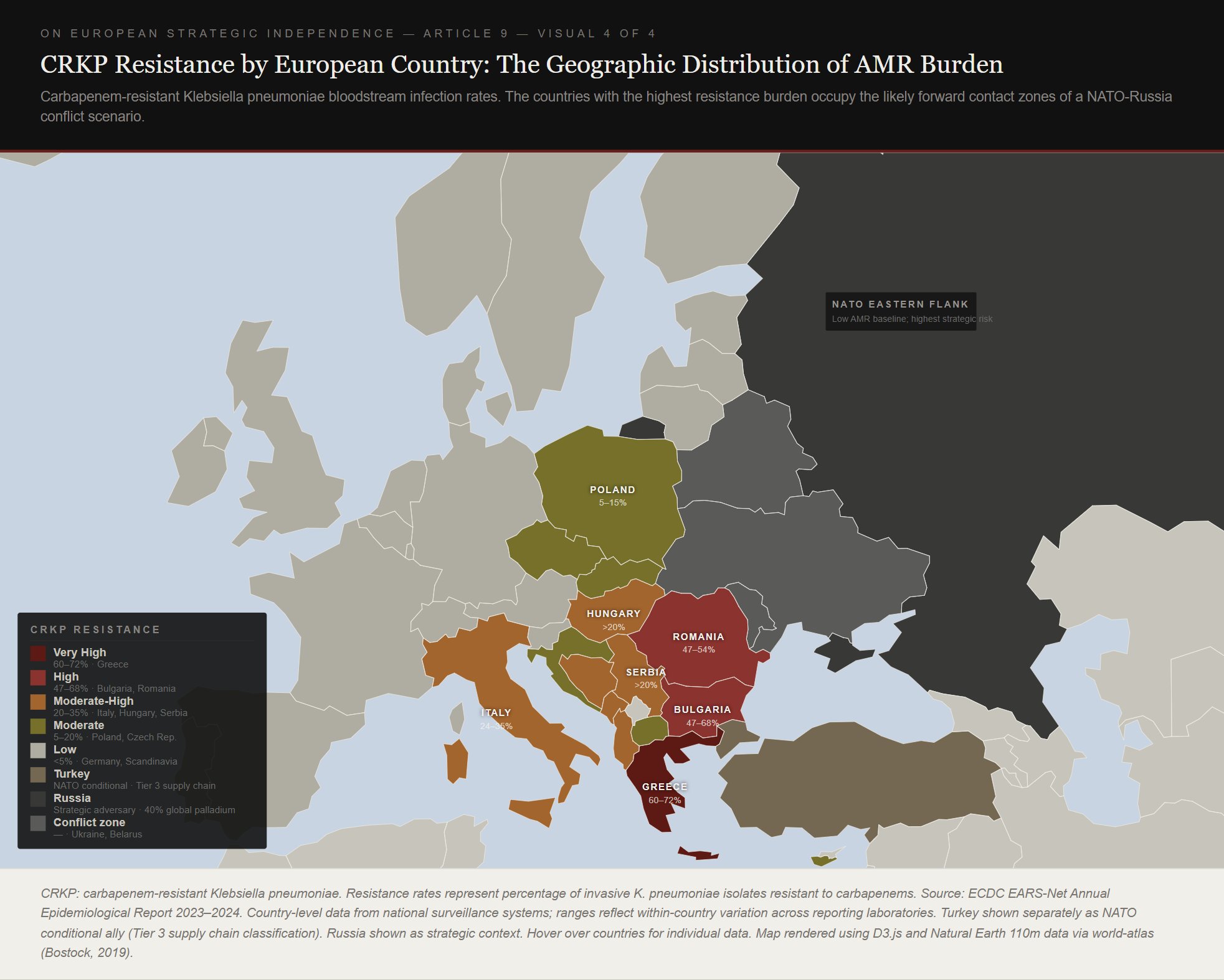
The map above makes visible something that aggregate European planning tends to obscure. The countries with the highest resistance burden, Greece, Romania, Bulgaria, and Italy, are also the countries whose forces would be concentrated on or near the eastern and southern flanks of any conflict scenario. The Baltic states and Scandinavia, which face the highest immediate military risk from Russia, have among the lowest resistance rates in Europe, under 8 percent. Coalition-level pharmaceutical planning that calculates average demand across all European forces conceals this asymmetry. The asymmetry matters because last-resort antibiotic demand is concentrated in precisely the forces in precisely the positions where resupply logistics are most constrained.
The problem deepens when European states expand from professional to conscript forces, which several have begun doing or are actively planning. Professional military forces are health-screened and receive regular medical care, and their resistance carriage is lower than general population averages. Conscript forces draw from the full population, including people with recent hospital contact in high-resistance healthcare systems.
A Romanian mobilization from 75,000 professionals to 300,000 conscripts brings the additional 225,000 from a general population where half of all serious Klebsiella infections are resistant to the most powerful standard antibiotics. Those additional soldiers are not individually more vulnerable than their professional counterparts, but the pharmaceutical system serving them must be prepared for approximately twice the last-resort antibiotic demand per wounded soldier compared to a German or Finnish equivalent. Within the limits of my research, that calculation appeared in no current European defense planning framework.
Applied to the worst-case scenario, a multi-front conflict beyond twelve months without US support, drawing on conscript forces from high-resistance populations, these conditions generate roughly 113,000 to 319,000 wound infections. Of those, between 11,000 and 48,000 will require last-resort antibiotics for organisms carrying the specific resistance gene, NDM-1, that the drug most European planners identify as the last resort does not actually cover.
That last sentence is where the treatment ladder becomes essential.
A reader might reasonably note that these figures assume European forces face the same supply failures Ukraine experienced, and that if Europe built adequate supply infrastructure the numbers would look different. Both observations are correct. But the supply infrastructure does not yet exist, and the coverage floors described below are precisely what this article argues must be built. Until they are, the figures from a supply-constrained scenario are the ones that matter for planning, not those from a scenario in which the problem has already been solved.
The treatment ladder for serious gram-negative wound infections, infections caused by a category of bacteria that includes Klebsiella, Pseudomonas, and Acinetobacter, runs through four steps.
Step 1 covers the majority of infections in the first 24 to 72 hours, before laboratory results are available, using standard broad-spectrum antibiotics like ceftriaxone and piperacillin-tazobactam. Step 2 escalates to carbapenems, the most powerful class of standard antibiotics, of which meropenem is the most widely used, when Step 1 fails or when the organism is already known to be resistant. Step 3 covers infections resistant to carbapenems, using newer antibiotic combinations such as ceftazidime-avibactam, where avibactam is a compound that protects the antibiotic from being broken down by the resistance enzymes the bacteria produce. Step 4 covers organisms that defeat Step 3, specifically, bacteria carrying the NDM-1 gene, which produces a resistance enzyme that avibactam cannot inhibit. The only drugs with documented clinical activity against these organisms are cefiderocol and a combination of aztreonam with avibactam.
The coverage thresholds at each step, 65 percent at Step 1, 80 percent at Step 2, 90 percent at Step 3, 100 percent at Step 4, are derived from the clinical cost of gaps at each level.
At Step 1, alternatives exist and surgical wound management can partially substitute, but the threshold is set at 65 rather than 50 percent because below 65 percent, inadequate dosing actively accelerates resistance in the treated population, pushing Step 1 patients up into Steps 3 and 4. At Step 2, the mortality curve steepens sharply below 75 percent coverage, and the 80 percent threshold sits above that inflection point, with a structured protocol using older, more toxic agents absorbing the remaining 20 percent. At Step 3, one toxic alternative exists, colistin, an older antibiotic with serious kidney side effects, which carries a documented 25-percentage-point higher thirty-day mortality compared to the preferred treatment. The 90 percent threshold allows a pre-planned colistin protocol for 10 percent of patients, which is clinically defensible where an unstructured shortage is not. At Step 4, the threshold is 100 percent because no effective alternative exists.
These thresholds carry a second purpose alongside the clinical one. The production architecture described later in this article spreads supply across multiple geographically separate facilities so that losing one site does not bring the whole system below the survival floor. For that architecture to work, each threshold must sit high enough above the minimum level at which patient deaths become unavoidable that the system can absorb the loss of one facility and still meet it. The clinical rationale and the resilience rationale are both present in these numbers. Understanding why the thresholds are set where they are requires seeing both.
A patient presenting with a pan-resistant NDM-producing organism who does not receive cefiderocol or aztreonam-avibactam does receive no care at all. The 100 percent target does not mean every patient survives, it means all available supply is administered through a structured rationing protocol that allocates it to the patients with the highest probability of surviving, rather than lost to unstructured shortage.

The critical limitation of ceftazidime-avibactam, the drug that most European planners would identify as the last resort for resistant infections, must be stated plainly, that it does not cover NDM. The pharmacological reasoning is that NDM produces a resistance enzyme that works by a different chemical mechanism than the one avibactam is designed to block. Against NDM-producing Klebsiella pneumoniae, ceftazidime-avibactam achieves less than 10 percent probability of reaching therapeutic levels in the bacteria regardless of dose or how slowly it is infused. For the 80 percent of infected war wounds carrying NDM-1 in the Ukrainian patient populations studied this drug does not work.
The actual last resort for NDM infections is cefiderocol, a drug that uses an entirely different mechanism, essentially smuggling itself into bacteria by mimicking the iron molecules they need to survive, bypassing the resistance pathways that defeat other antibiotics. It is manufactured by a single company globally, Shionogi of Osaka, Japan. Global annual production is estimated at between 0.1 and 0.5 tonnes. In the worst-case conflict scenario, based on my estimates and calculations, European forces would require between 1.75 and 7.34 tonnes.
These figures apply the triage thresholds to patient volume projections derived from the Ukraine war reference data, adjusted for worst-case force size and resistance escalation over the duration of a multi-front conflict. The worst case requires cefiderocol at between fifteen and seventy-three times estimated current global annual production, from a single manufacturer, for the resistance pattern that 80 percent of infected war wounds carry. That is the number against which everything that follows in the supply chain analysis should be read.
The Layer Below the Layer
The European pharmaceutical manufacturing that exists is, in significant parts, a final assembly operation for inputs that come from somewhere else entirely. The facility is European, but the supply chain that feeds it frequently is not. Under the conditions that make the supply chain matter most, a European address offers no protection against the disruption of the inputs a facility requires.
Step 1 and the 7-ACA problem
Ceftriaxone, pronounced sef-try-AX-one, is the standard first antibiotic given to seriously wounded soldiers within the first three hours of injury, as recommended by US and NATO combat wound protocols. It is the entry point to the treatment ladder. At the 65 percent triage coverage threshold against worst-case demand, Europe would need between 1.06 and 2.98 tonnes of ceftriaxone active ingredient. There is one European manufacturer certified to produce it at the required quality standard, ACS Dobfar in Trivolzio, northern Italy. The rest of global supply comes from Aurobindo Pharma in India, which exports approximately 1,200 tonnes annually, and from Chinese manufacturers.
ACS Dobfar's production of ceftriaxone requires 7-ACA, 7-aminocephalosporanic acid, as its chemical foundation. This compound is to ceftriaxone the core structure onto which everything else is assembled, like a pre-formed steel frame of a building. Without it, production cannot proceed, and there is no European production of 7-ACA. The last European manufacturer, Corden BioChem in Germany, ended production around 2020 under Chinese price competition, a fact documented by the American Chemical Society. Every gram of 7-ACA used in European ceftriaxone production, including at ACS Dobfar, is imported from China.
Ceftazidime, the Step 3 antibiotic, shares the same upstream dependency on 7-ACA. Cefiderocol, the Step 4 last resort, requires the same chemical foundation. Three of the four steps in the treatment ladder trace back to a single upstream void with no European production of the chemical that all three drugs require.
Step 2 and the final assembly problem
Meropenem is the primary carbapenem, the most powerful class of standard antibiotics, used at Step 2. For infections with highly resistant organisms, the dosing is 6 grams per day for 14 days, producing a per-patient requirement of 84 grams. Against worst-case demand at the 80 percent coverage threshold, Europe would need between 0.93 and 2.61 tonnes of meropenem active ingredient. ACS Dobfar produces this at its Trivolzio facility.
The meropenem synthesis process combines two chemical intermediates and then strips away their protective chemical groups using palladium, a precious metal, as a catalyst.
The first intermediate, the carbapenem bicyclic core, the most structurally complex component of the molecule, known in the industry as MAP, has no meaningful European manufacturer. The global supplier list for MAP consists of Xinxiang Juyuan Biological Technology, Shaanxi Dideu Medichem, and Ningbo InnoPharmChem. All three are Chinese specialty chemical manufacturers, and without MAP, ACS Dobfar's meropenem synthesis stops, regardless of the investment in the facility or the capability of its staff.
The second intermediate, a sulfur-containing compound that determines the antibiotic's range of activity, is also produced exclusively by Chinese manufacturers. Made-in-China.com lists 364 suppliers of meropenem-related intermediates in China. The synthesis of this intermediate uses DMF, dimethylformamide, an industrial solvent whose primary European producer is BASF at its Ludwigshafen complex, a facility cluster I will revisit and examine in article 10 of this series.
The palladium catalyst required in the final synthesis step comes 40 percent from Russia's Nornickel mining company and 40 percent from South Africa's Bushveld mining complex. Russia is both the adversary in the conflict scenario and a critical input supplier for the synthesis of the drug most urgently needed to treat that conflict's wounds.
One further connection belongs here because it runs directly to the previous article in this series. The protective chemical groups used in meropenem's MAP intermediate are derived from a compound that traces upstream to toluene, a basic industrial chemical produced in oil refineries and chemical cracker plants. Article 8 established that European TNT production for artillery shells depends on toluene from the same European chemical facilities that are closing under Chinese price competition. Artillery shells and meropenem synthesis compete for inputs from the same contracting industrial base.
Turkey, and the borosilicate vial problem
Every injectable antibiotic in the treatment ladder is delivered through a glass vial, Type I borosilicate glass, a chemically resistant formulation that does not react with the drugs it contains and can withstand sterilization temperatures. Both US and European drug safety regulators require it for injectable medicines, and no substitute is approved. Type I borosilicate glass requires 12 to 15 percent boron trioxide in its composition, which comes from borate minerals, of which Turkey controls 72 to 75 percent of global reserves through Eti Maden, a state enterprise.
European vial manufacturers are genuinely world-class. Schott AG in Germany holds approximately 35 percent of global pharmaceutical vial market share. Gerresheimer AG, Stevanato Group in Italy, and SGD Pharma in France cover most of the rest of European supply. The vial manufacturing industry is a European strength, but the upstream raw material it depends on is different.
In 2022, Russian disruption of European gas supplies forced Schott AG to reduce output at its German plants by 12 to 15 percent and increased vial prices 20 to 25 percent for pharmaceutical clients. Borosilicate glass production requires furnaces operating at between 1,400 and 1,650 degrees Celsius, powered primarily by natural gas. The conflict scenario that generates maximum demand for injectable antibiotics is the same scenario that disrupts the gas supply that vial production requires.
Schott has responded to the Turkish boron concentration by investing in boron mine equity in Argentina, which is the right strategic instinct, applied at one company's level at a fraction of the required scale. A genuine European response would require either developing European boron deposits, minor deposits exist in Saxony and the Czech Republic but are not currently in commercial production, or coordinated investment across the European vial manufacturing industry into non-Turkish boron sources in Argentina and the United States. Neither of these exist as a program, but both are necessary.
Step 3, and the Pfizer concentration
Ceftazidime-avibactam is the primary drug at Step 3, and its clinical limitation must be restated here because it determines what Step 3 actually covers, that it does not work against NDM. The avibactam component blocks a specific class of resistance enzyme, serine beta-lactamases, but NDM produces a different class of resistance enzyme, a metallo-beta-lactamase, that avibactam cannot inhibit. The drug that covers KPC-producing and OXA-48-producing resistance, two other important carbapenem resistance mechanisms, is clinically inactive against the NDM resistance pattern documented of infected war wounds.
This matters for the supply chain analysis because the commercial rights to ceftazidime-avibactam outside the United States belong to Pfizer, acquired from AstraZeneca in 2016. The same transaction gave Pfizer commercial rights to meropenem distribution and aztreonam-avibactam development rights. A single deal placed commercial control of the drugs covering 80 to 100 percent of the triage requirement at Steps 2, 3, and 4 in the hands of one US-headquartered company.
The structural exposure this creates has been described in the reframing section. A possibility for how to respond to this exists in principle, but has never been used in practice. The TRIPS Agreement, the international treaty governing intellectual property rights for pharmaceuticals, contains Article 31, which permits governments to authorize production or procurement of a patented medicine without the patent holder's consent when national security or public health emergency requires it, in exchange for reasonable compensation to the patent holder.
EU member states have this right individually, as there has been no European-level compulsory licensing framework established calibrated specifically to activate under conflict conditions for strategically critical medicines. This instrument does not solve the production problem, since Europe would still need to build manufacturing capability, but it resolves the commercial rights dependency that would otherwise prevent Europe from legally producing or procuring these drugs from non-Pfizer sources even if the capability existed. Establishing that instrument is just as necessary as building the production infrastructure.
Step 4 and the cold chain problem
Cefiderocol is unique in the treatment ladder in several ways, and the combination of them is more significant than any one individually.
It is the only drug with documented activity against NDM-producing pan-resistant organisms, the specific resistance profile documented in the Ukrainian patient populations studied. It is manufactured by a single company globally, Shionogi of Japan. Shionogi's own regulatory filings identify an inability to build production capacity to meet demand as a formal product risk, in peacetime, for a drug approved in the EU since 2020.
No public production figure exists for cefiderocol. The range of 0.1 to 0.5 tonnes annually is inferred from the drug's single-manufacturer status and its market reach since EU approval in 2020. The uncertainty in this estimate does not change the structural conclusion, since even at the upper end of the range, the worst-case demand figure is more than three times total global output. Worst-case European conflict demand is estimated between 1.75 and 7.34 tonnes.
The molecule's structure is also unique. Cefiderocol uses bacterial iron transport systems to enter cells that have closed their normal entry channels against other antibiotics. It essentially mimics the iron that bacteria need to survive, smuggling the antibiotic inside by exploiting the bacteria's own nutrient uptake mechanism. This mechanism is what makes it active against NDM organisms.
It also means its synthesis requires a specific class of chemical precursors, catecholamide siderophore compounds, that trace upstream to phenol chemistry, which depends on benzene from the same European chemical cracker facilities I will examine in the next article. The drug is connected to the industrial contraction at the heart of this series through its own specific chemistry.
The minimum timeline from a technology transfer agreement with Shionogi to European production approved by the European Medicines Agency is seven to ten years. Most assessments place Russian military readiness for large-scale conventional operations in the 2027 to 2030 window, with 2028 being cited most often.
European cefiderocol production cannot be achieved within the threat window regardless of when the decision is made, as the chemistry, the regulatory pathway, and the manufacturing infrastructure cannot be compressed below seven years on any realistic timeline. The decision must still be made now, begin building a stockpile from current Shionogi production immediately, open technology transfer negotiations with Shionogi immediately, and commission a European production program in full knowledge that it will not arrive in time for the immediate threat window but that the medium-term requirement is permanent.
One further detail about cefiderocol is a logistics problem with a specific structural implication. Every other drug in the treatment ladder can be stored at room temperature. Cefiderocol powder for infusion must be continuously refrigerated between 2 and 8 degrees Celsius. A strategic stockpile requires refrigerated military storage facilities with backup generator power.
Under conflict conditions that target energy infrastructure, and Russia has conducted more than 1,800 documented attacks on Ukrainian health facilities, a refrigerated pharmaceutical depot is simultaneously a known military asset and a system that electricity disruption can render useless. The drug most urgently required at the end of the treatment ladder is the only one whose strategic stockpile is vulnerable to the energy infrastructure disruptions that a conflict scenario produces. That combination is a structural condition the planning framework must acknowledge.
Three connections the supply chain reveals
Three cross-cutting findings emerge from the analysis that the drug-by-drug account cannot fully convey.
Russia appears not once but three times in the pharmaceutical supply chain. It is the primary source of 40 percent of global palladium, which is required as a catalyst in producing meropenem and avibactam. It is the origin of the gas supply disruption that reduced European pharmaceutical glass vial production 12 to 15 percent in 2022. And it is the adversary whose conflict operations would simultaneously maximize demand for these drugs and constrain the inputs on which their production depends.
The fermentation process used to produce penicillin G, which is the starting point for the amoxicillin and first-generation antibiotic chain, requires nitrogen sources derived from industrial ammonia. European industrial ammonia production contracted dramatically under energy price pressure. Yara, one of Europe's largest fertilizer producers, operated at approximately 35 percent of capacity during the 2022 energy crisis, and Grupa Azoty at approximately 10 percent.
In article 11 I will document that the same nitrogen chemistry contraction threatens European food security through fertilizer production shortfalls. The fermentation that produces first-line battlefield antibiotics and the agricultural system that produces European food compete for the same nitrogen chemistry base under the same energy pressure from the same structural cause.
Artillery shells and meropenem synthesis compete for toluene from European chemical cracker plants that are closing. In article 8 I established that ammunition and pharmaceuticals share upstream chemistry concentrated in adversarial territory. The toluene connection adds a layer within the pharmaceutical supply chain itself, as the medicine that treats the wounds and the shells that cause them draw from the same contracting industrial base.

The visual above makes the pattern visible across the full treatment ladder simultaneously. The Tier 1 columns, production under genuine European control, thin out rapidly as the analysis moves from finished dose toward raw materials. Most Tier 3 dependencies represent a single external source that Europe does not control. For meropenem and cefiderocol, the problem is compounded. Multiple independent vulnerabilities reinforce each other at the same time. Chinese intermediates, Russian palladium, Turkish boron for the vials, and US commercial rights over distribution all apply simultaneously to the same drugs. The visual marks these cases separately as Tier 3 with compounding vulnerabilities, meaning that no single investment closes the gap.
What sovereignty would require
Building genuinely redundant European pharmaceutical sovereignty for the treatment ladder, covering production, intermediate chemistry, raw material supply, energy independence at each production node, hardened storage, and logistics infrastructure, runs to between 6.8 and 14.8 billion euros across a 7 to 10 year program. The lower figure reflects the minimum needed to establish working production capability at each rung of the treatment ladder. The higher figure reflects what the worst-case planning standard actually requires, which is multiple production sites in different geographic locations, each hardened against attack and capable of operating independently if another is lost.
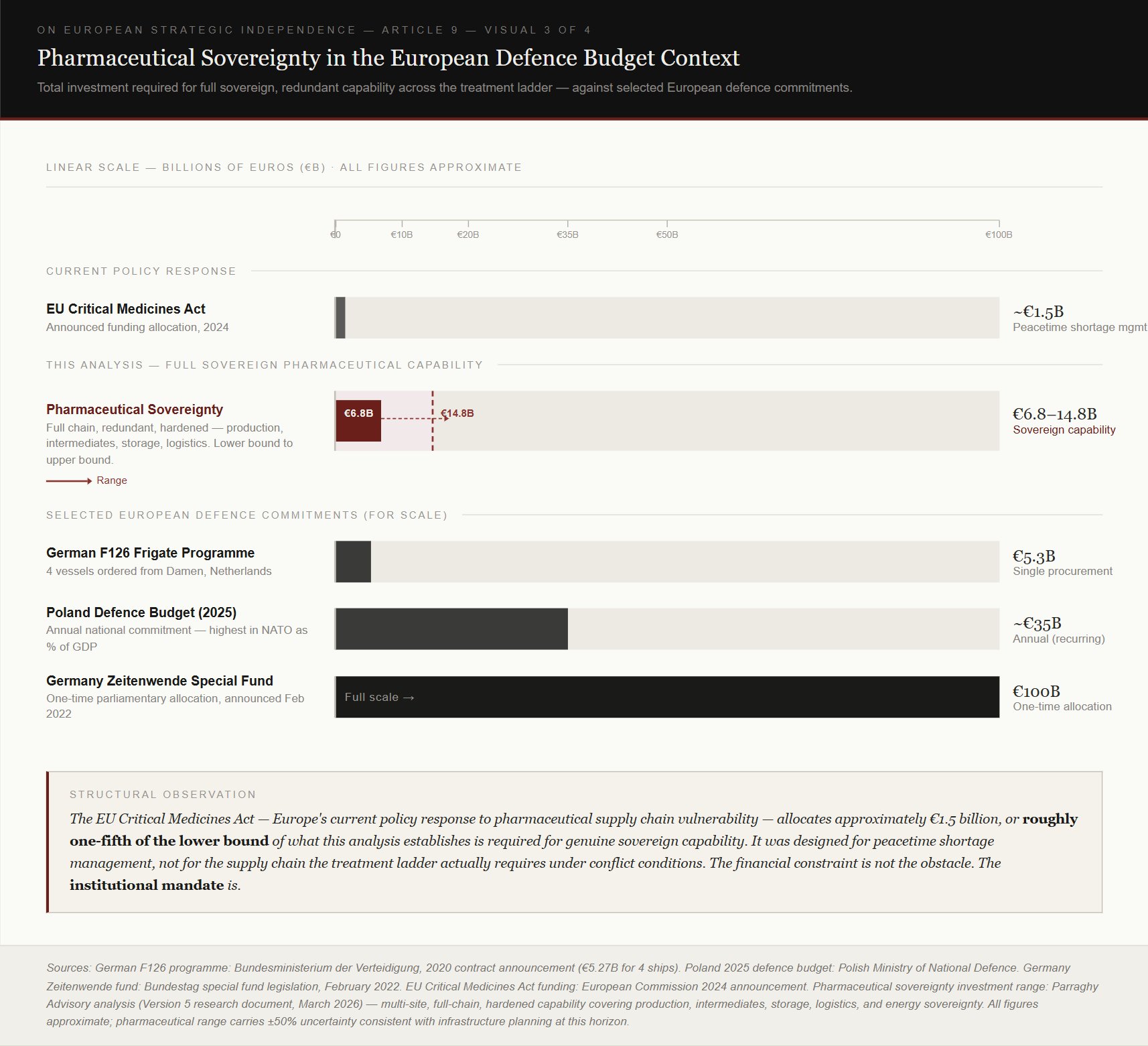
The visual above places that figure against selected European defense commitments. Germany's Zeitenwende special fund, announced in February 2022, was 100 billion euros, a one-time parliamentary allocation. Poland's annual defense budget for 2025 is approximately 35 billion euros, the highest in NATO as a share of GDP. The F126 frigate program, four warships ordered from Damen Shipyards, costs approximately 5.3 billion euros. The pharmaceutical sovereignty program required to treat the casualties of the forces those commitments exist to field costs less than two F126 programs at its lower bound. The institutional constraint is what has prevented this from being created before.
No European authority currently has the mandate to commission a classified pharmaceutical manufacturing program. The European Defense Agency handles defense procurement. HERA, the Health Emergency Preparedness and Response Authority, handles health emergency preparedness. Neither has authority over the other's domain. Neither can legally commission a hardened military pharmaceutical production facility. The EU Critical Medicines Act, Europe's current policy response to pharmaceutical supply chain vulnerability, was designed for peacetime shortage management, procuring more of what the healthcare system currently uses, through commercial markets. It allocates approximately 1.5 billion euros, or roughly one fifth of the lower bound of what genuine sovereign capability requires. And it was designed for the wrong problem, as the treatment ladder under conflict conditions requires qualitatively different drugs, at volumes that do not exist in Europe, from supply chains whose upstream dependencies run through adversarial territory.
The security classification argument must be stated plainly, including a point that standard parliamentary oversight framing obscures. The transparency requirements governing pharmaceutical manufacturing, facility certification, drug master file submissions, location disclosures, are appropriate and necessary for peacetime pharmaceutical governance and civilian use. They become a targeting database when applied to facilities whose primary strategic function is wartime pharmaceutical sovereignty. Facility locations, production capacities, raw material suppliers, and supply chain architectures are publicly accessible through the regulatory filings that pharmaceutical manufacturers must submit. A hybrid warfare campaign designed to collapse European healthcare capacity before a military engagement does not need to destroy these facilities. It needs to know where they are and what they depend on, and the transparency framework provides that.
The solution is a structure where two systems operate side by side. Civilian pharmaceutical quality oversight continues under existing transparency rules, as it should. The military reserve production capability operates under a classified framework, following the model that already governs nuclear facilities, intelligence operations, and special forces programmes in every functioning democracy. Budget decisions about the program remain subject to parliamentary approval in the normal way. What is restricted is the operational information that would allow an adversary to identify where the facilities are, what they produce, and what they depend on.
This distinction matters because a different approach has already demonstrated its limits in concrete terms. Parliamentary security committees across Europe carry the formal requirements for handling classified material, but parliamentary rules in most European systems create disclosure obligations that apply when questions are raised in parliament or in committees, in some cases, regardless of the sensitivity of the subject.
In Germany, far-right Alternative for Germany (AfD) lawmakers have submitted thousands of parliamentary inquiries about German military and intelligence. The inquiries focused on drone defenses, weapon transports, and critical infrastructure. This reflects a genuine disagreement about where democratic transparency ends and operational secrecy begins.
In others, it reflects the presence of individuals representing political movements whose relationship to the constitutional order is contested. The AfD's presence in German parliament and on various committees has made this concern concrete and visible. The effect on operational security is the same regardless of what drives it. Sensitive information about military capability has entered wider circulation through the very mechanism designed to provide accountability for it.
The model this article proposes is not the removal of democratic oversight, because parliamentary approval of the budget remains. What changes is that the operational aspects and specifics of the infrastructure are handled the same way a navy handles a submarine's patrol routes. Oversight rests with vetted individuals within the professional security and military chain, not with members of parliament and committees whose membership rotates with electoral outcomes and whose rules were written for a political environment where every major party could be assumed to accept the constitutional order.
Multiple geographically separated production sites are the final structural requirement, a precondition of its strategic value. A single production facility, regardless of its cost, is a single point of failure. A precision strike, a cyberattack on the industrial control systems managing fermentation or chemical synthesis, or a sustained energy disruption can remove it from the operational picture.
Genuine pharmaceutical sovereignty requires geographically distributed production across different threat profiles, with western European sites at strategic depth for primary production and strategic reserves, intermediate sites in Germany and the Netherlands for logistics and surge capacity, and hardened forward production in Poland for the highest-volume Step 1 agents, where resupply to Baltic forces otherwise runs through the 65-kilometre Suwalki corridor that is simultaneously the most likely point of Russian interdiction in any Baltic scenario.
That architecture is a defense planning problem, and it requires institutional authority that no current European organization possesses. What this analysis establishes is the minimum precondition. The next layer of the same question is whether the architecture itself would survive the conflict conditions it is built to serve, and that question belongs in a different article.
The pattern the article belongs to
The supply chain gaps traced in this article were produced by governance choices made over decades, in full visibility, by institutions with, in some instances, the information to have chosen differently.
Market competition frameworks that prioritized lowest-cost procurement over supply chain security allowed Chinese bulk chemistry to outcompete European production on cost, for the cephalosporin core, for meropenem intermediates, for a dozen other inputs that are now exclusively Asian. Alliance dependency assumptions substituted nominal partnership structures for physical production capability, treating NATO membership or EU candidacy as a proxy for supply chain reliability without asking whether conflict conditions would keep those relationships aligned. Regulatory transparency requirements designed for peacetime pharmaceutical governance were applied without modification to infrastructure with military operational significance, because no institutional framework existed that could distinguish between the two.
And the separation between defense procurement and healthcare policy, which is appropriate for ordinary governance, became a structural obstacle to the investment program that sits precisely at the boundary of both.
These are the same governance design choices that produced European energy dependence on Russian gas, European ammunition production shortfalls, and the semiconductor vulnerabilities that a separate series of crises has made visible. The pharmaceutical supply chain is one expression of a structural miscalibration that runs across the entire industrial base, with governance frameworks designed for a world of economically interdependent partners with converging interests, applied without revision to a world in which strategic competition among states with partially opposed interests has reasserted itself as the operative condition.
Articles 10 and 11 will trace the same pattern through the bulk industrial chemistry and nitrogen supply chains that pharmaceutical production, agricultural production, and military production all draw from simultaneously. The steam cracker that closes under Chinese price competition removes a shared upstream resource from three supply chains at once. The nitrogen chemistry contraction that threatens fertilizer production also constrains antibiotic fermentation. The toluene unavailable for TNT is also unavailable for meropenem intermediates. These are the same structural conditions expressed at different levels of the industrial stack.
What makes the pharmaceutical case worth examining in this kind of detail is that it produces the clearest statement of what the failure costs. A government that sends its citizens into conflict accepts an obligation to ensure that no soldier dies from a preventable cause that the state had the capacity to address and chose not to. The supply chain analysis is the account of how far European governance currently is from meeting that obligation, because the institutional architecture required to act on it has never been established.
Sources:
- ECDC EARS-Net Annual Epidemiological Reports 2023-2024
- Holubnycha and Kholodylo, Communications Medicine, Nature Publishing Group, September 2025 (n=73 Ukrainian wounded)
- Schultze et al., EuroSurveillance, 2023 (cross-border NDM-1 transmission, Netherlands)
- USGS Mineral Commodities Summary 2024 (palladium production by country)
- JRC Palladium Impact Assessment (EU palladium supply security)
- PMC 2023, peer-reviewed process chemistry review of meropenem synthesis
- RSC Organic and Biomolecular Chemistry, 2020 (avibactam synthesis route)
- EMA Fetcroja Summary of Product Characteristics (cefiderocol cold chain requirement)
- Sandoz official communications, March 2024 (Kundl production capacity)
- C&EN (Chemical and Engineering News), American Chemical Society, 2020 (Corden BioChem cessation of 7-ACA production)
- Pfizer press release, 2016 (AstraZeneca acquisition of small molecule anti-infective portfolio)
- Shionogi regulatory risk disclosures (cefiderocol production capacity)
- Korean CRE Treatment Guidelines, 2024 (colistin vs ceftazidime-avibactam mortality data)
- US Joint Trauma System, Combat Wound Infection Prevention and Management, CPG ID:24
- Siboni and Bazak, "The IDF “Victory Doctrine”: The Need for an Updated Doctrine", Jerusalem Institute for Strategy and Security (JISS)
- World Trade Organization, TRIPS Agreement, Article 31